💌 Issue 143: ENDOSURE gets CE mark | Septagen launches to tackle cardiovascular risk | Osteoboost raises $8m
The global weekly briefing on women's health innovation and Femtech
Welcome to issue #143 FutureFemHealth, (w/c April 20 2026) — the global weekly briefing on women’s health innovation.
🌟 In this week’s briefing:
🦴 Osteoboost raises $8M for first FDA-cleared wearable targeting bone loss
❤️ Septagen targets overlooked cardiovascular risk in women with precision biotech platform
✅ ENDOSURE secures CE mark for 30-minute endometriosis test that can be used from teenage years through to post-menopause
👩🏽💻 UK strategy refresh lands - as US women’s health research funding falls 31%
Share your news: anna@futurefemhealth.com
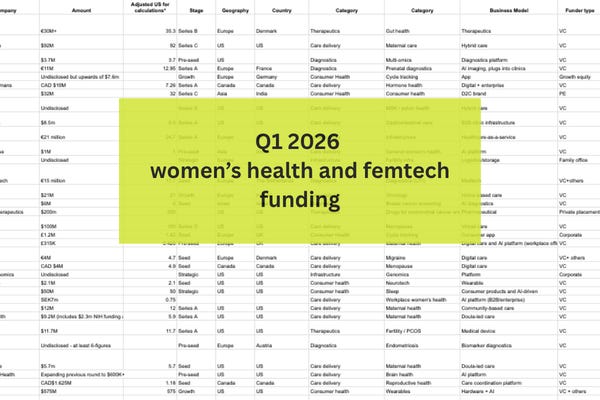
But first…Q1 in review
Women’s health raised $1.24 billion in Q1 - but nearly half came from a single deal (WHOOP). Strip that out and the Q1 figure drops to $665.
In case you missed it, in our first FutureFemHealth Pro feature I break down five key signals - from the rise of doula care to the shift towards deeper tech like menstrual blood diagnostics, and what it means for where the market goes next.
Companies like gastrointestinal care and migraine are serving majority populations - often 80% women - without being labelled as femtech.
If we don’t track this properly, a large part of women’s health innovation stays invisible.
Read the full breakdown - available inside FutureFemHealth Pro - and download our complete Q1 funding tracker here:
💰 Capital flows
📌 US: Osteoboost raises $8M to scale FDA-cleared wearable targeting bone loss in postmenopausal women. The prescription device delivers vibration therapy to the hips and spine and is positioned as the first non-drug treatment for osteopenia - the precursor to osteoporosis and a stage where there are currently no approved pharmaceutical options. This round was led by Ambit Health Ventures. (Continue reading: FutureFemHealth)
📌 THE NETHERLANDS: ScreenPoint Medical raises €13.6M to scale AI-powered breast cancer detection platform. The Transpara platform supports radiologists in identifying cancers earlier and prioritising high-risk cases, and is already deployed in 30+ countries. It’s another example of AI embedding into clinical workflows - not replacing clinicians but acting as a ‘second pair of eyes’ to improve accuracy and efficiency. €11.9 million of the round came from existing investors Insight Partners and Siemens Healtineers, with a further €1.7 million in non-dilutive grants. The company will use funding to support product innovation and global expansion. (Continue reading: EU-startups)
🌟 Industry moves and strategic shifts

📌 US: Septagen launches to develop a drug and diagnostic platform targeting cardiovascular risk in postmenopausal women. Cardiovascular disease is the leading cause of death in women, but treatment has largely remained one-size-fits-all and often based on male physiology. This new approach from a Vanderbilt University-spinout targets thromboinflammatory risk - a platelet-driven mechanism linked to estrogen decline - using a blood test to identify women at elevated risk, followed by a once-daily oral drug designed to address that specific pathway alongside existing treatments. While still preclinical, it could bring precision medicine and a more targeted approach to an area of women’s health that’s long lacked specific therapies.(Continue reading: FutureFemHealth)
📌 EU: ENDOSURE receives CE mark for a non-invasive endometriosis test designed to deliver results in 30 minutes. This test can be used across all ages, including adolescents and postmenopausal women - extending beyond existing diagnostics which often limit to ages 18–43. With diagnosis still often delayed for years - and reliant on invasive procedures like laparoscopy - the company is positioning this as a first-line tool that could enable earlier detection and intervention, particularly in younger patients where the disease often begins. Clinical validation and real-world performance is still evolving - and will be key to determining the role of tests like these in practice. (Continue reading: FutureFemHealth)
📌 GERMANY: Hello Inside is partnering with insurer Barmer to offer a metabolic health programme to over 9 million members. Bringing continuous glucose monitoring into mainstream care, users will track their blood sugar alongside lifestyle factors, with AI translating that data into personalised recommendations. The company is applying this specifically to women’s health - across areas like PMS, menopause and energy - where underlying metabolic issues are often missed. (Continue reading: Tech.eu)
📌 US: Progyny launches a fully insured fertility and women’s health plan for small employers, moving beyond its traditional large, self-insured client base. The new fixed-cost model aims to lower the barrier to offering fertility benefits and open access to a much larger segment of the workforce. Small employers represent a significant untapped segment in the US, with around 50 million people working in organisations that have historically lacked access to fertility benefits. It moves fertility on from being just a premium perk at the largest companies to something that could be available as standard. (Continue reading: Fierce Healthcare)
📌 US: Wisp partners with Visby Medical to offer FDA-authorized at-home PCR STI testing with integrated telehealth follow-up. This model combines fast, lab-quality diagnostics (results in ~30 minutes) with immediate access to treatment, shortening the time from testing to care into a single at-home pathway. While not entirely new, it’s a good example of how women’s health is now starting to benefit from more integrated test, diagnose and treat models that reduce friction for patients. This is especially important in areas like sexual health where speed, privacy and stigma are still key barriers to seeking out help. (Continue reading: Med City News)
📌 EU: FertilAI secures CE mark for AI tools designed to optimise timing decisions during fertility treatment cycles. The Fertilane platform integrates into clinic workflows and uses data from 100,000+ cycles to predict ovulation and oocyte yield, helping clinicians determine the optimal moment for intervention. The focus on timing targets one of the most complex and variable parts of fertility care - while also helping clinics manage capacity and reduce monitoring burden. It signals growing use of AI not just for diagnosis, but for real-time decision support within treatment pathways. (Continue reading: FutureFemHealth)
📌 First confirmed pregnancy using BAIBYS’ AI system that selects and isolates sperm in IVF. This is a first-in-human milestone for fully automated sperm selection - because while AI tools are already used in IVF to support sperm and embryo selection, these systems typically guide embryologists rather than replace them at this part of the process. The procedure was carried out in Paris in December 2025, involving a 37-year-old patient, with the pregnancy now confirmed beyond the first trimester. (Continue reading: FutureFemHealth)
📊 From FutureFemHealth Intelligence
📌 NEW REPORT: The women’s health wearables landscape. Wearables in women’s health are arguably still ‘early’ - but so is the system they’re trying to improve. As devices move beyond fitness into areas like menstrual health, menopause and pelvic care, they’re beginning to generate something that has historically been missing: continuous, real-world data on women’s bodies. This report, written by women’s health strategist Anastasiya Markvarde, maps the emerging landscape from head to toe. (Continue reading: FutureFemHealth)
🩸 Research and women’s health news
📌 UK/AUS: New Lancet review finds “brain fog” affects two-thirds of women during menopause - yet remains under-researched and poorly understood. Cognitive changes are a major area of unmet need in menopause research. Symptoms like memory loss and reduced concentration impact confidence, work and day-to-day functioning for many women, but we don’t know why or what types of support or treatment could be effective. Importantly, symptoms are also hard to quantify or measure - which means it’s easier for the system to ignore or dismiss them. “I hear all the time from women who have gone through menopause that validation would have made a significant difference to their resilience and the approach they took to living with menopause.” says lead researcher Dr Caroline Gurvich (Continue reading: UCL / The Lancet)
📌 CANADA: Survey of 1,000 women finds gaps in reproductive health knowledge, diagnosis and care have remained unchanged across generations. Women across age groups are still experiencing near identical challenges in access to care — suggesting that despite growing attention on women’s health, the system itself has not meaningfully improved. More than half of respondents said their symptoms were initially dismissed, while over 60% needed multiple providers to get answers. The findings point to a broader issue beyond access: a lack of information and preparation, with over half saying sex education did not prepare them for adult reproductive health and many delaying care because they were unsure their symptoms were serious enough. (Continue reading: FutureFemHealth)
📄 Policy watch: risks and opportunities
📌 UK: England’s Women’s Health Strategy to include £1.5m femtech fund as refresh focuses on patient voice and waiting lists. The long-awaited update of the original 2022 women’s health strategy came with some blunt honesty from Health Secretary Wes Streeting as he said the NHS “too often gaslights women, treating their pain as an inconvenience and their symptoms as an overreaction.” A small £1.5m Femtech fund (with limited detail so far) sits within a wider push to support tech adoption - but may point to the immediate priority being cultural and system change instead. Other notable points include a requirement for publicly funded research to account for sex-based differences, and plans to trial wearables in areas of deprivation, initially focused on cardiovascular disease. (Continue reading: FutureFemHealth)
📌 US: Scientific research funding in the U.S. is shrinking, according to an analysis by The Washington Post. Analysis of NIH grant data is showing that ~15% fewer competitive grants were awarded and more than 2,700 fewer projects funded so far this year. Women’s health has been hit particularly hard, with a 31% drop in funded projects, while labs face hiring freezes, reduced PhD admissions and early-career researchers struggling to secure funding. The impact here is long-term, creating a weaker pipeline that will have knock-on impacts for years to come. (Continue reading: Washington Post)
📌 US: Perelel founder pushes for benefits change for postpartum support. Around one-third of Americans utilize HSA/FSA benefits to subsidize their spending on approved goods (which includes things like the Oura Ring and infrared sauna blankets). Yet, the tax code largely covers a woman’s health only before giving birth, an underlying disconnect between government regulation and women’s health standards. Perelel founder Alex Taylor is campaigning to make postnatal vitamins HSA- and FSA-eligible, in the same way that prenatal vitamins already are. (Continue reading: Fortune)
📆 Save the date
📌 VIRTUAL: Menopause 2.0 ‘Understanding perimenopause before menopause’. May 6. Women of Wearable’s third annual virtual conference on menopause, this year featuring speakers from Natural Cycles, Amissa Health, Mayo Clinic and more. 🎟️ Use code ‘FUTUREFEMHEALTH’ at the checkout for a free ticket here.
📌AUSTRALIA: Breaking the silos: the women’s health medtech summit. Sydney, May 27-28. This inaugural event is dedicated to addressing long-standing gaps in research, innovation, and policy affecting women’s health in Australia. The program brings together leading voices shaping the national conversation on women’s health in MedTech - spanning media, policy, research, and industry - to drive real system change. 🎟️ Tickets here.
That’s all for this week! If you’ve missed any previous newsletter issues catch them all at futurefemhealth.com and do make sure to follow us on LinkedIn and you can connect with me directly.
Anna
This newsletter is for informational purposes only and should not be considered medical or financial advice.







Great coverage this week! Love the Endosure news. And the wearable diagram made me laugh. We are going to be covered in wires. What if they start talking to each other and short circuiting? I see a rom-com in the works!